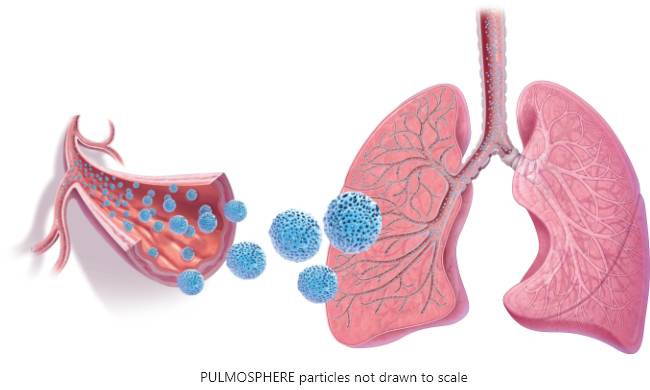
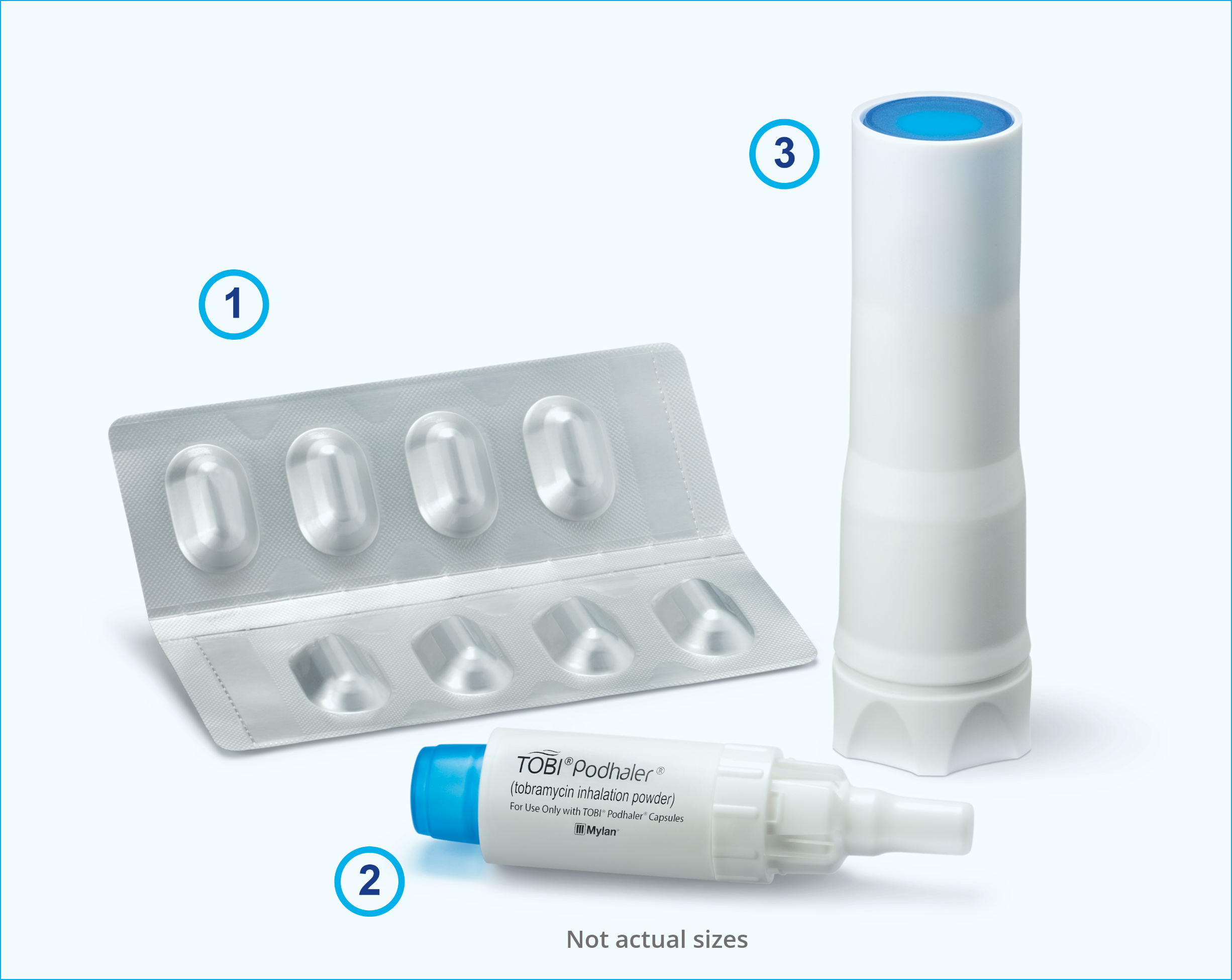
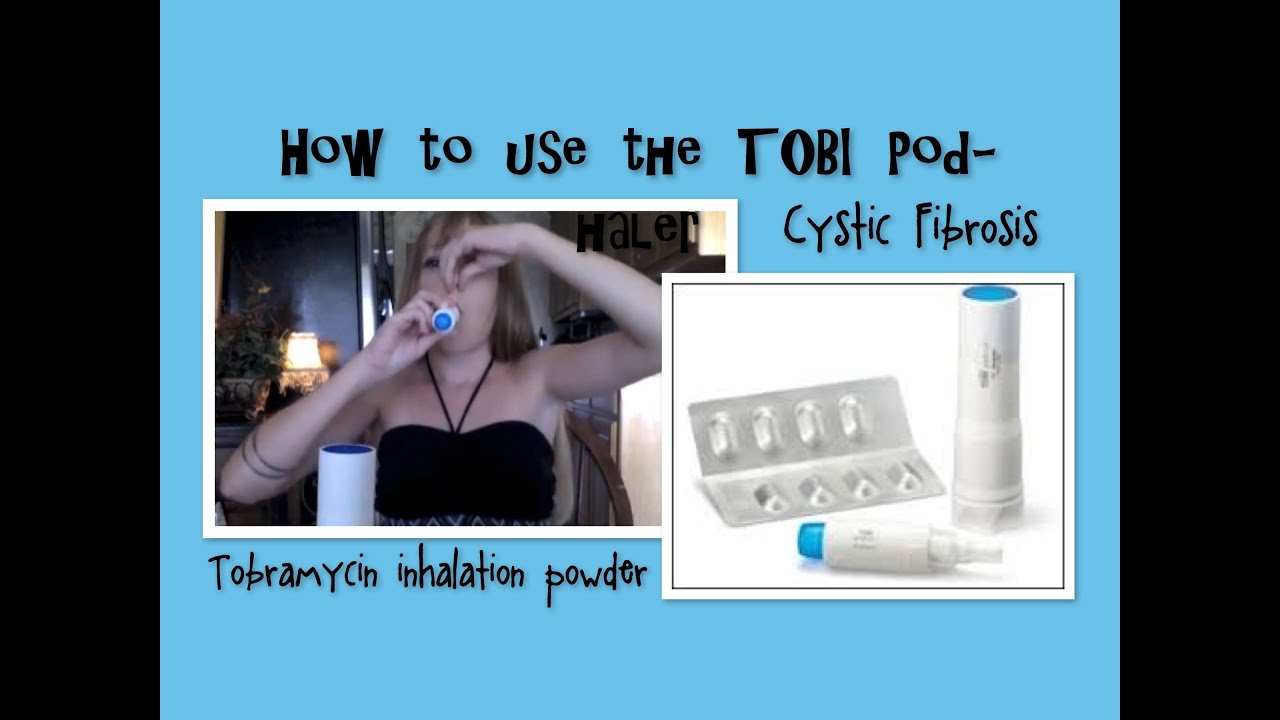
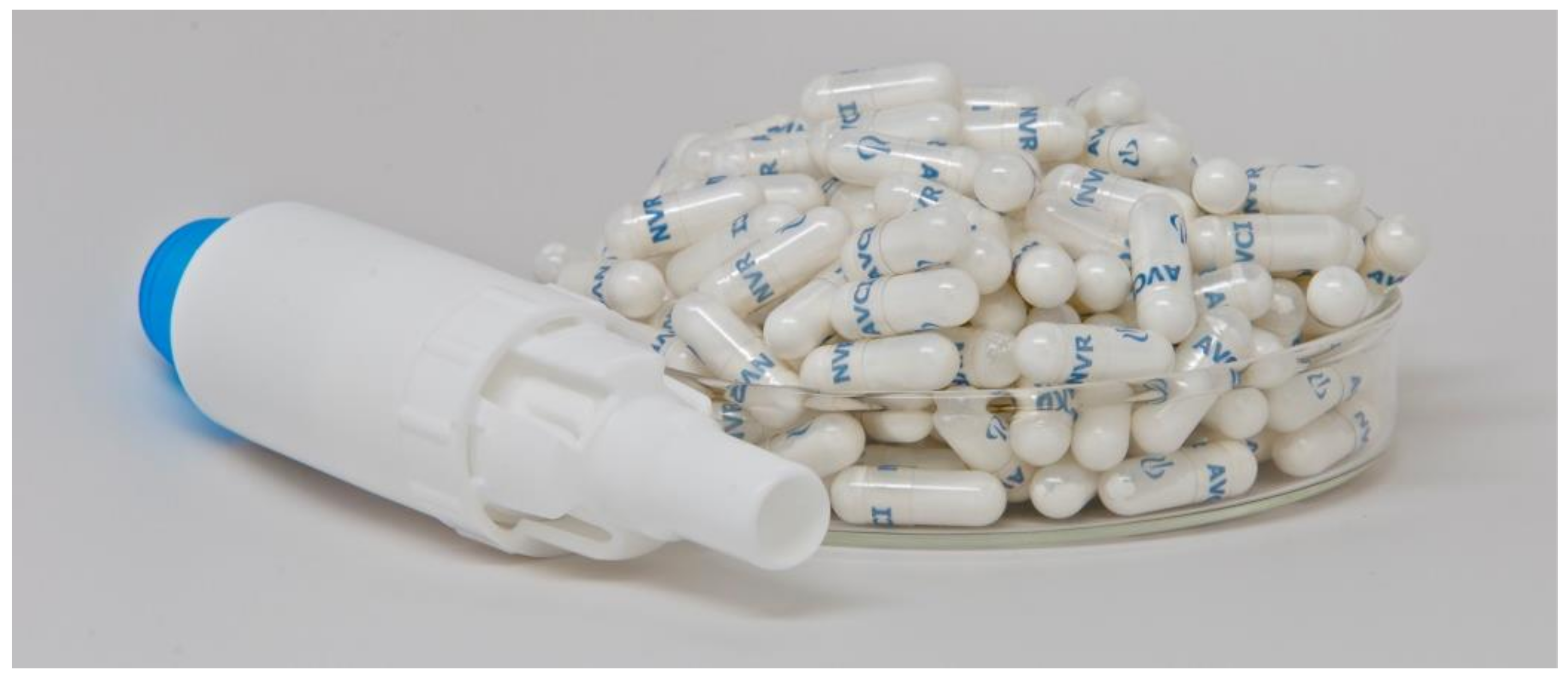
FDA approves Novartis TOBI® Podhaler™ for certain cystic fibrosis patients, the first and only dry powder inhaled antibacterial in US | Fierce Pharma

FDA approves Novartis TOBI® Podhaler™ for certain cystic fibrosis patients, the first and only dry powder inhaled antibacterial in US | Fierce Pharma
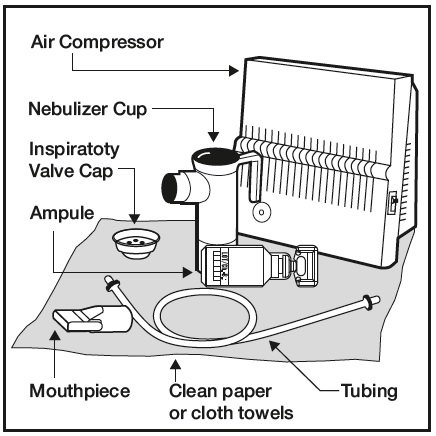

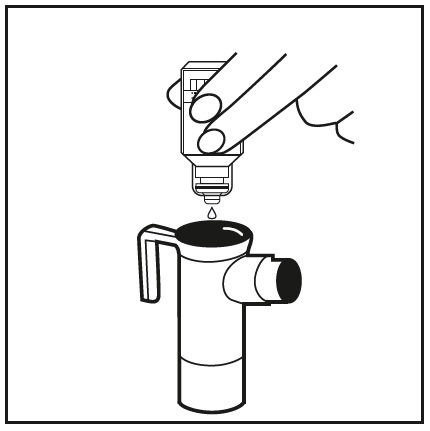

![Comparison of TOBI ® and PFIT, inhaled tobramycin products [3,4]. | Download Table Comparison of TOBI ® and PFIT, inhaled tobramycin products [3,4]. | Download Table](https://www.researchgate.net/publication/288855940/figure/tbl1/AS:391358515826690@1470318549076/Comparison-of-TOBI-R-and-PFIT-inhaled-tobramycin-products-3-4.png)