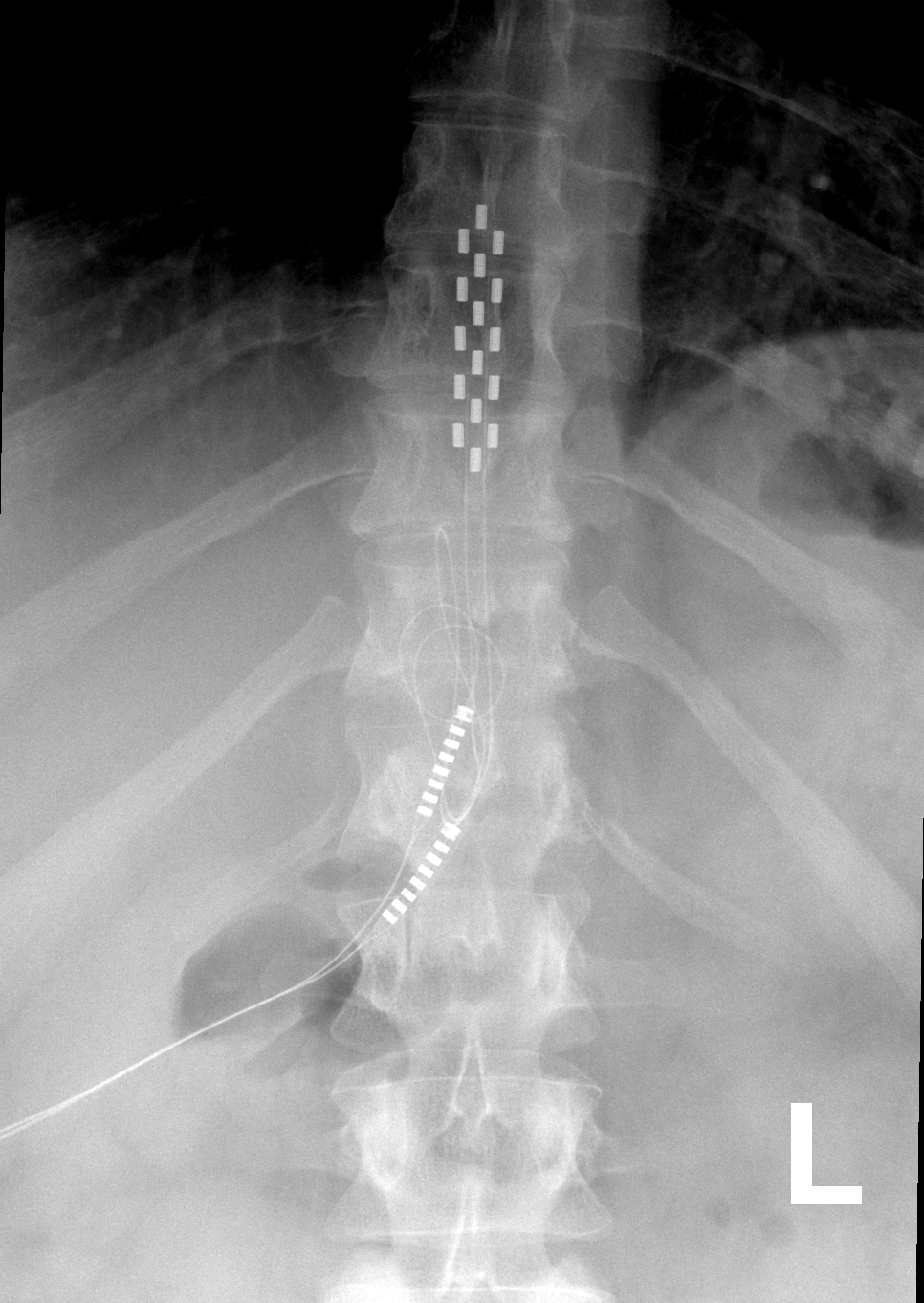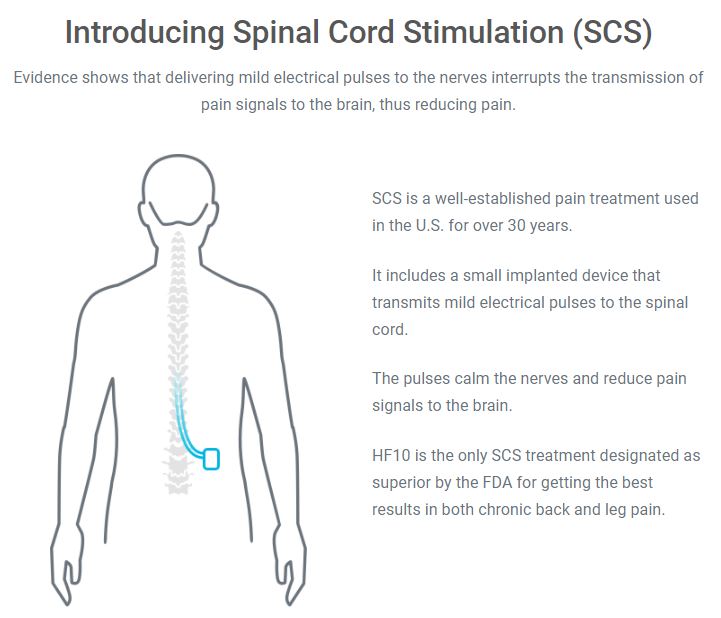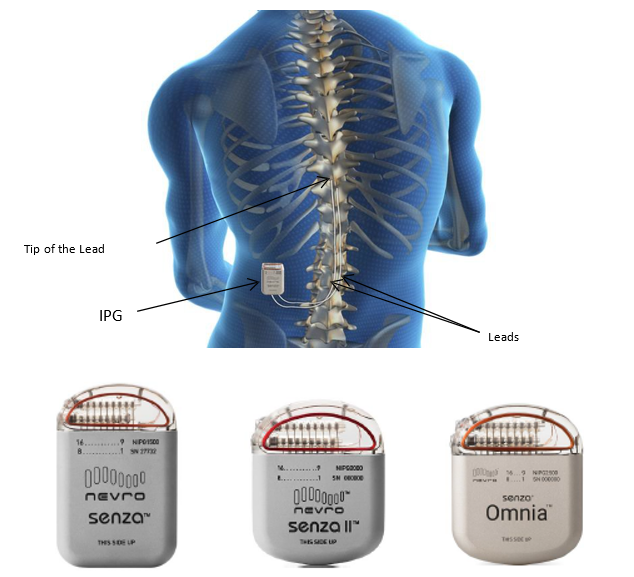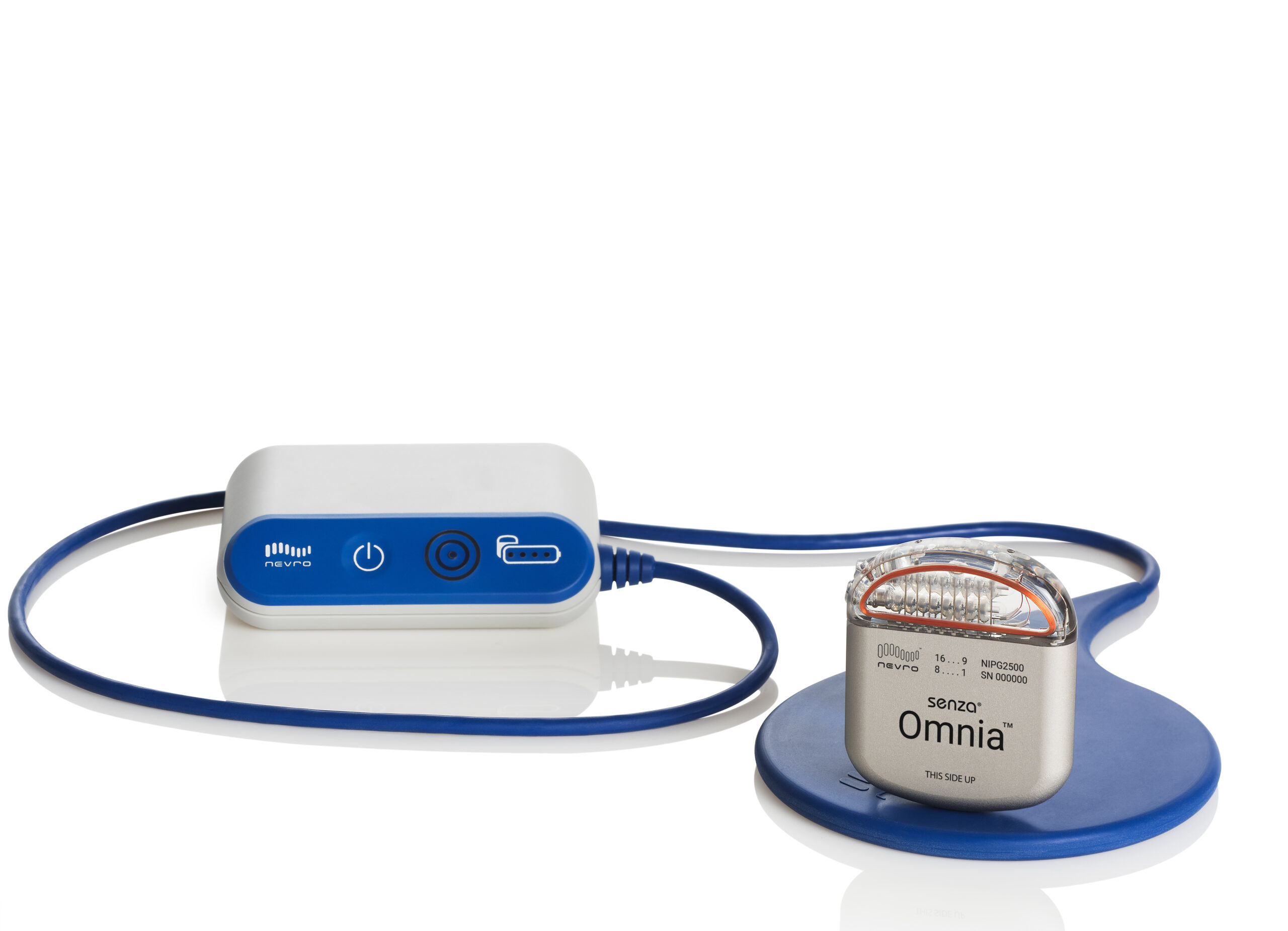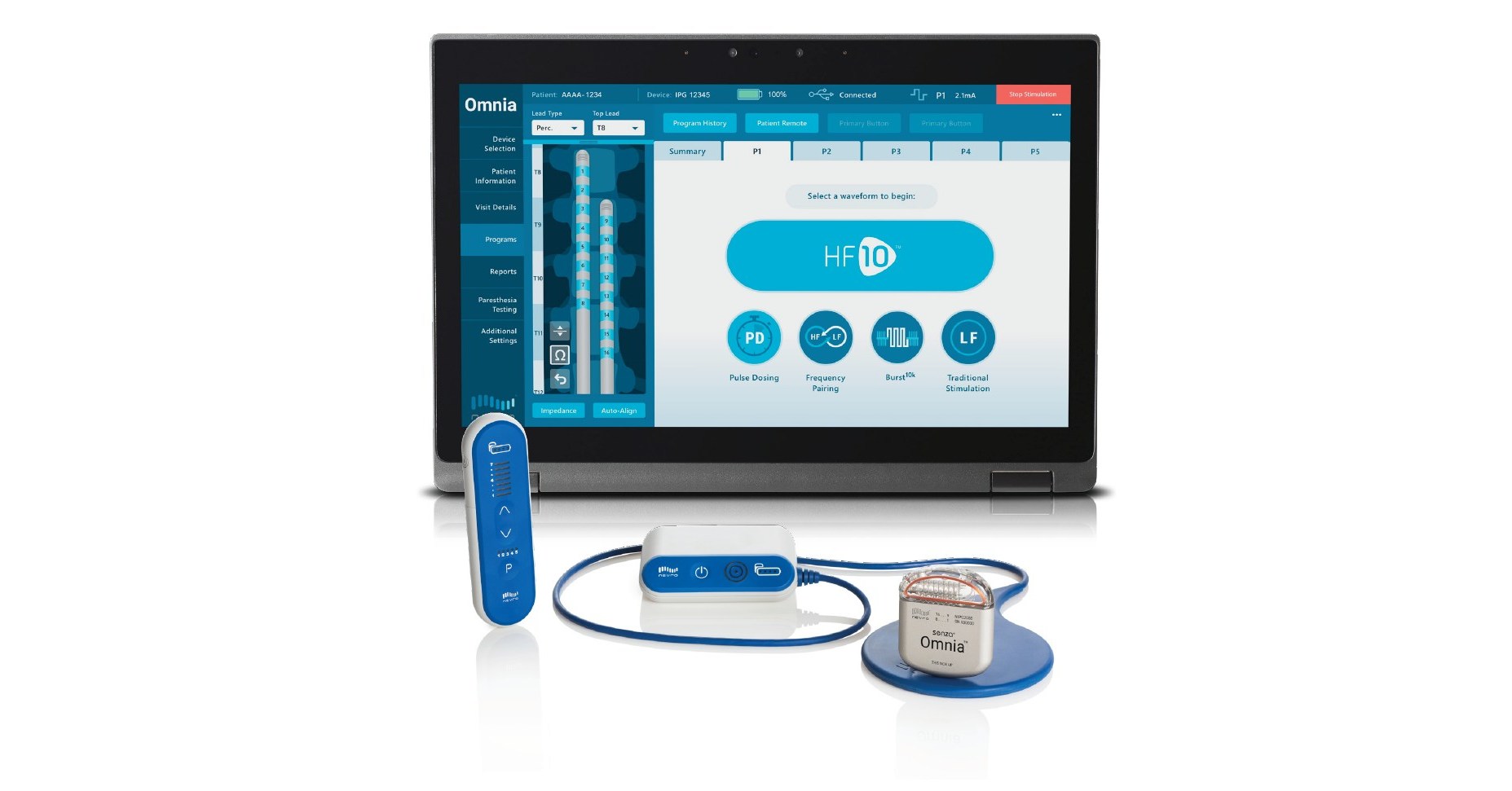
Nevro Announces CE Mark Approval of Senza® Omnia™ Spinal Cord Stimulation System to Treat Chronic Pain

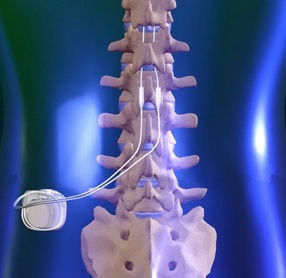
Spinal cord stimulation for the treatment of peripheral neuropathic pain | Tidsskrift for Den norske legeforening

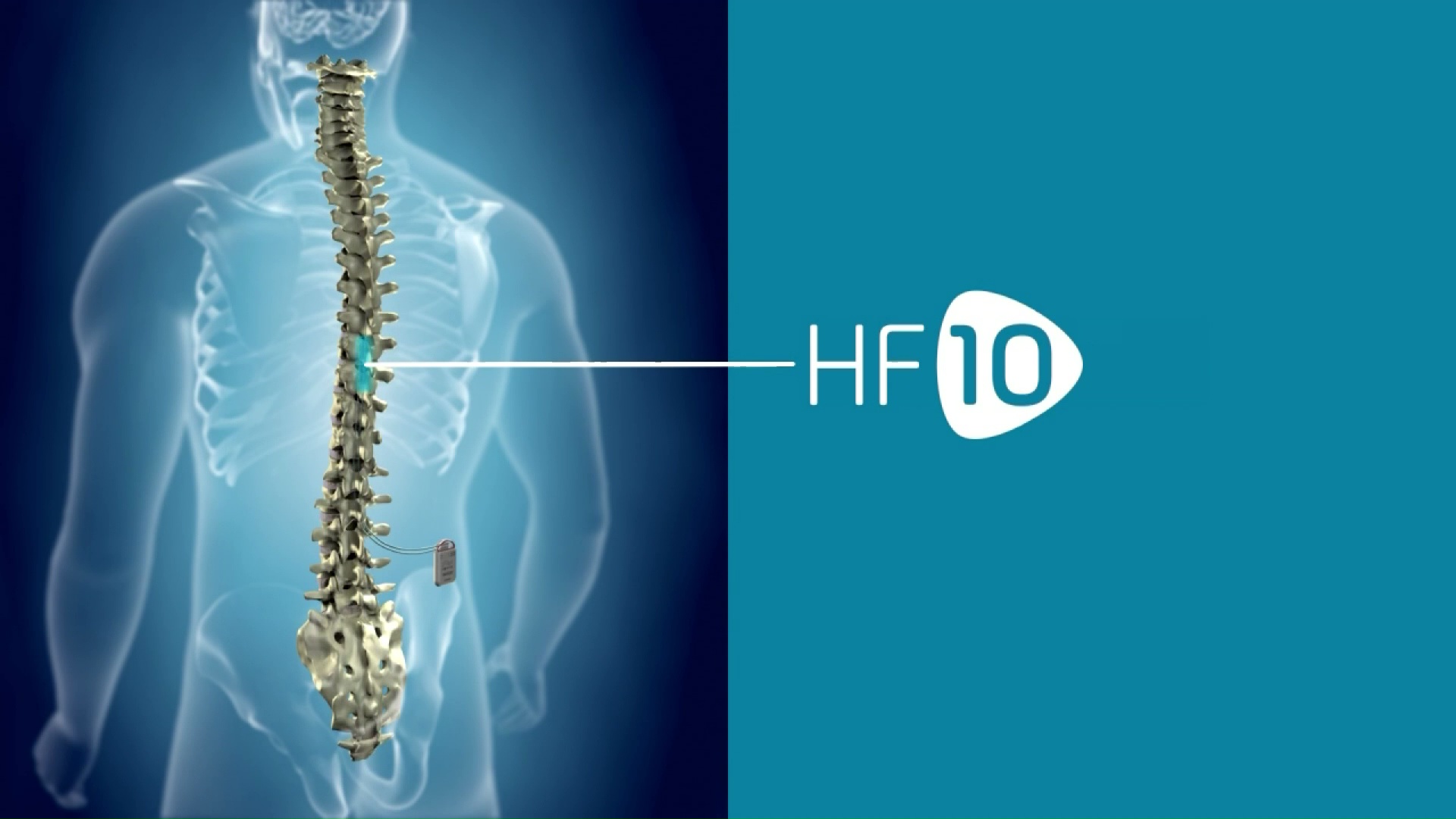
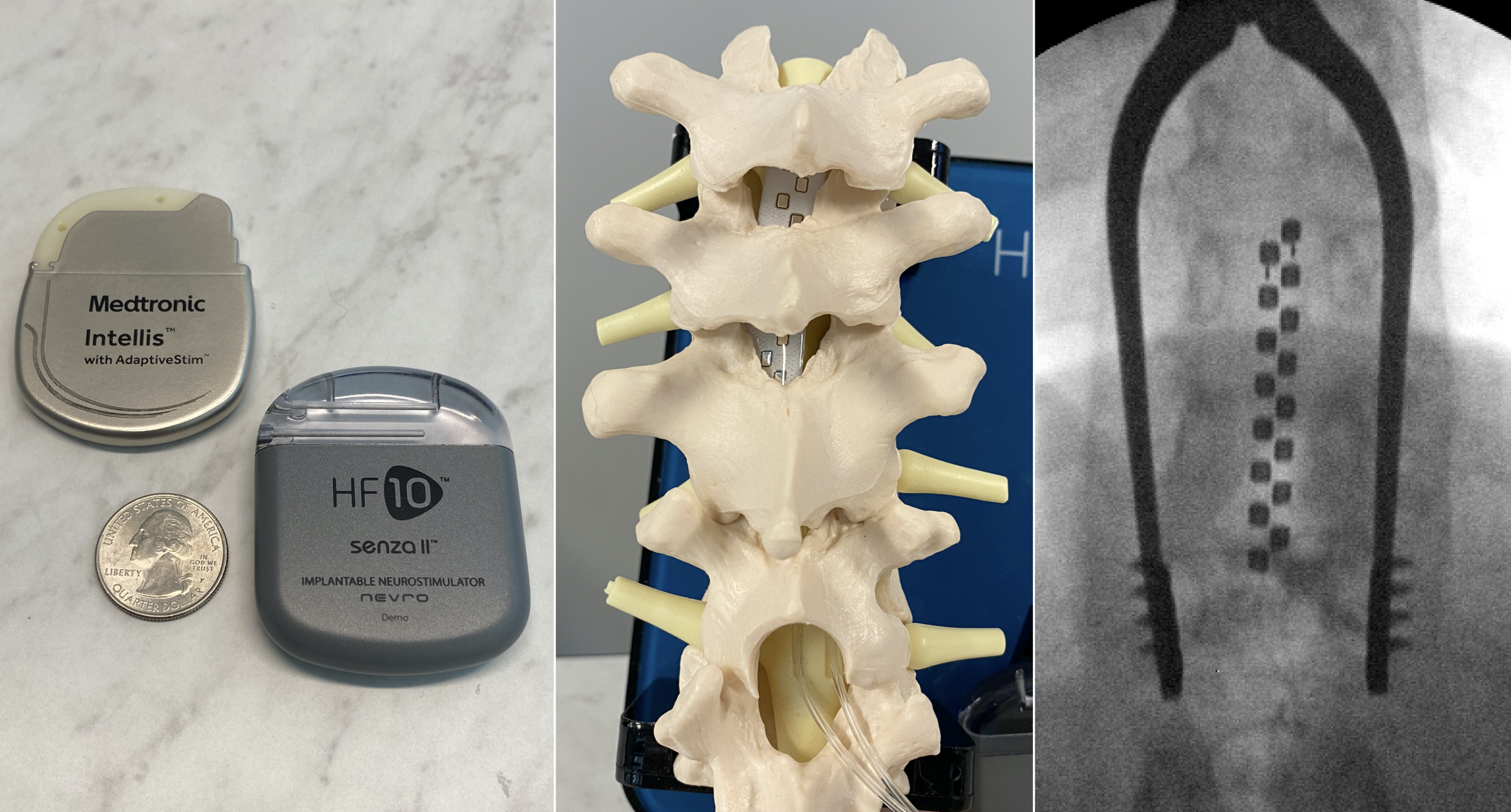
Nevro Announces FDA Approval of its 10 kHz High Frequency Spinal Cord Stimulation Therapy for Treatment of Chronic Pain Associated with Painful Diabetic Neuropathy (PDN)