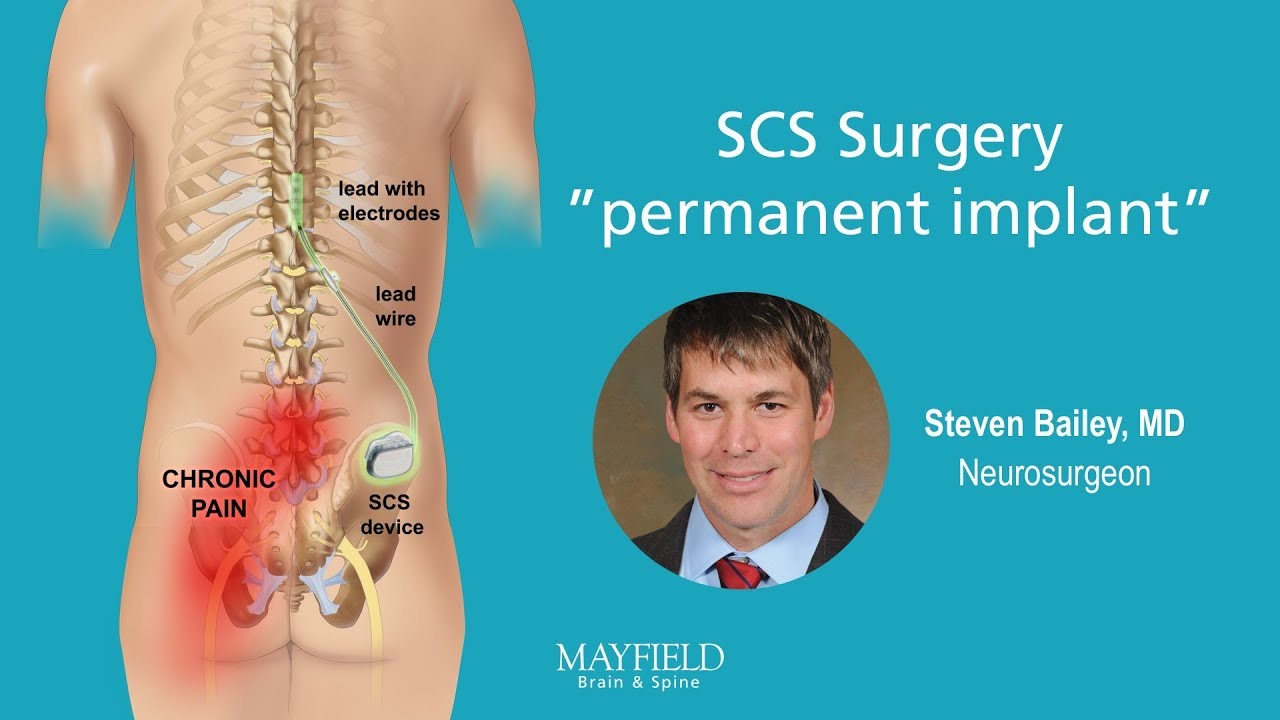
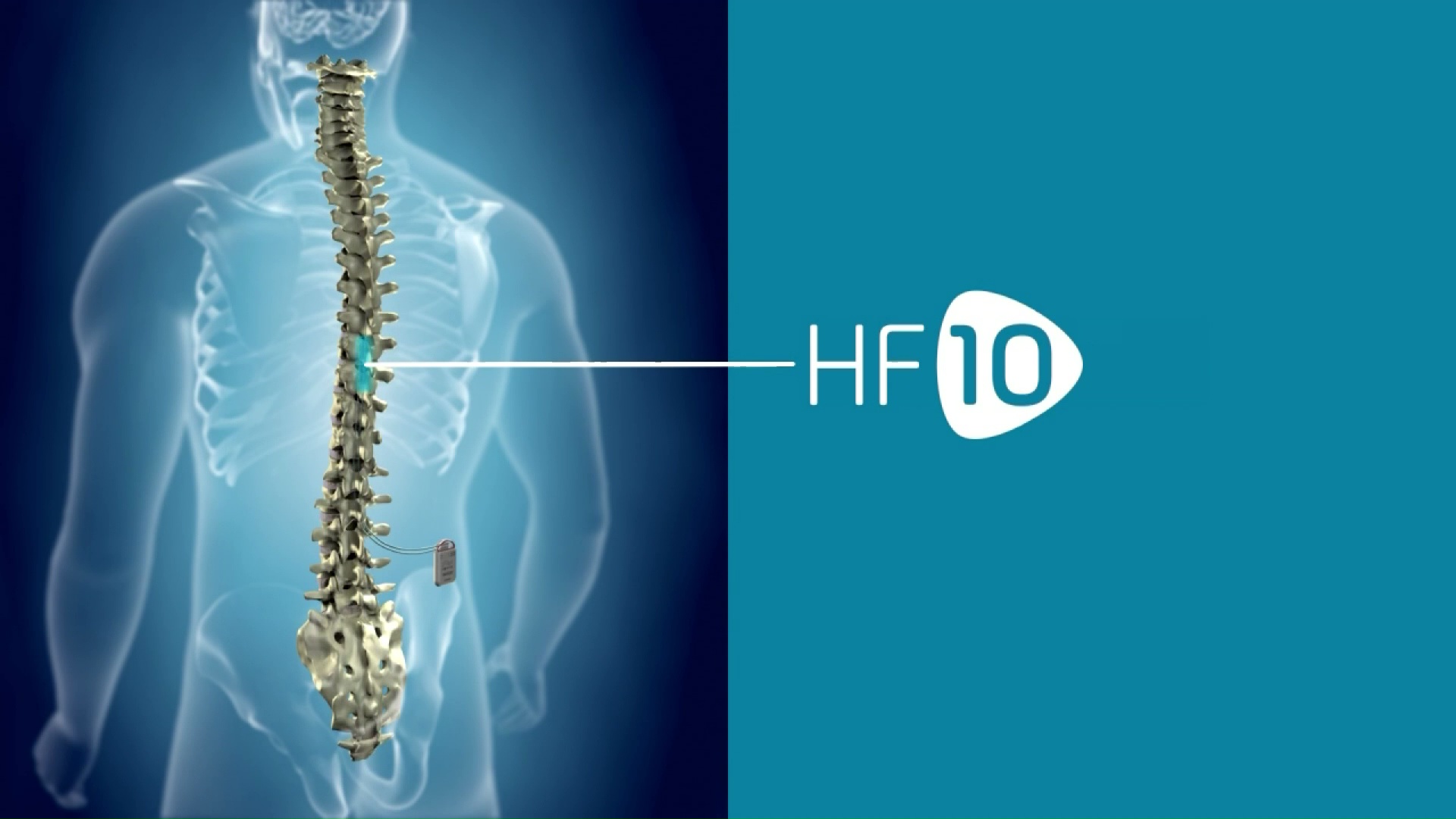
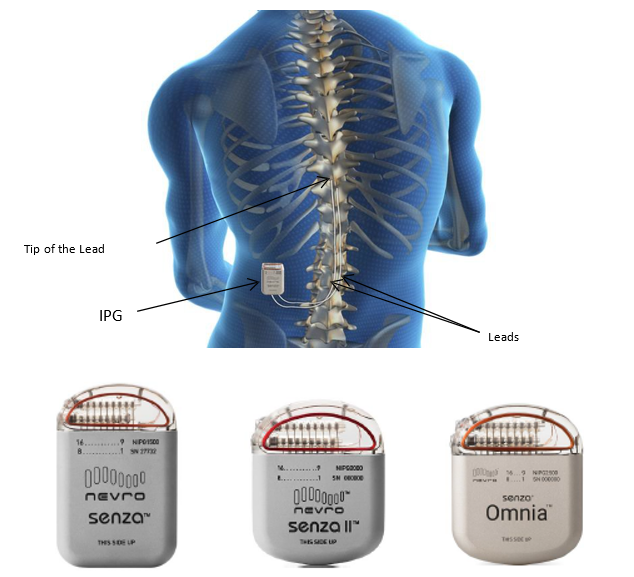
/assets/production/practices/5265d2f0ddd60c799633e9c35cafac7762ca2892/images/2602718.jpg)
High-Frequency Spinal Cord Stimulation for Newburyport, Danvers and Gloucester, MA | Pain Management Centers of New England | Pain Management

FDA approves Nevro spinal cord stimulation to treat chronic pain with diabetic neuropathy - Drug Delivery Business

Nevro Receives FDA Approval For Senza II Spinal Cord Stimulation System Delivering HF10 Therapy | Orthopedic Design Technology
Health Management and Leadership Portal | Implantable neurostimulator / medullary Senza™ Nevro Corp. | HealthManagement.org

Multicentre, double-blind, randomised, sham-controlled trial of 10 khz high-frequency spinal cord stimulation for chronic neuropathic low back pain (MODULATE-LBP): a trial protocol | Trials | Full Text