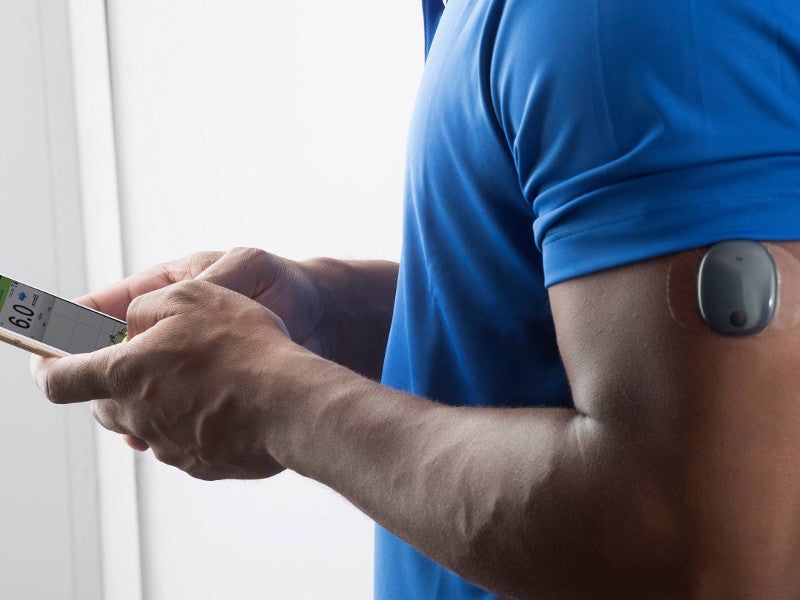
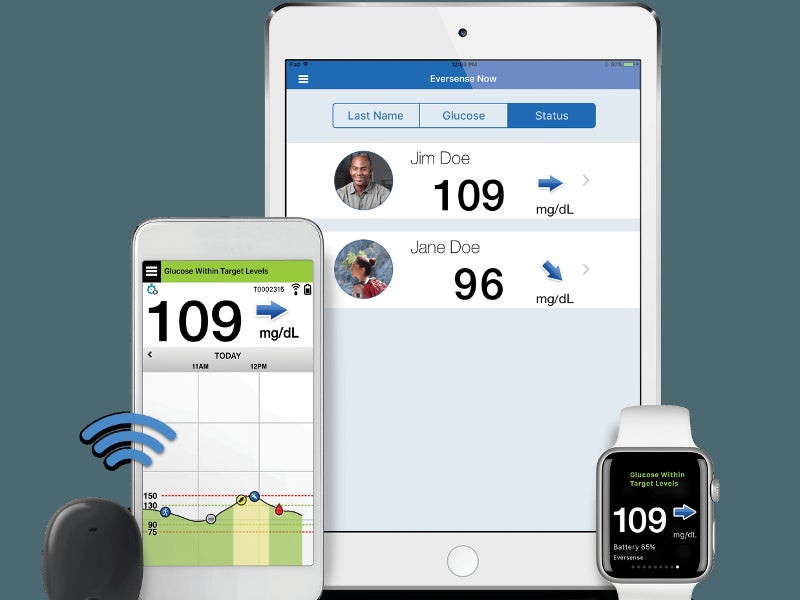
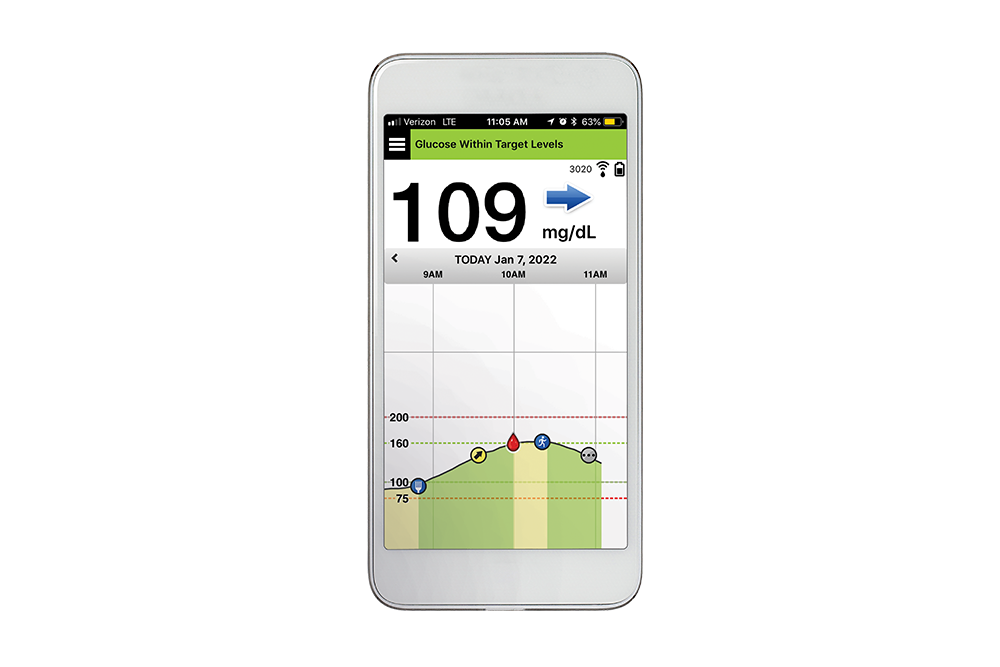
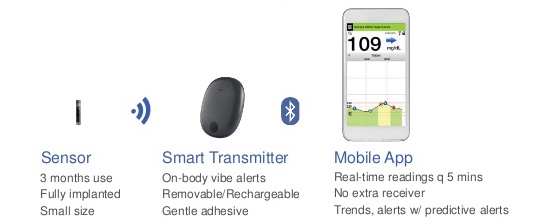
Eversense sensor placement. (a) The positioning guide for the Eversense... | Download Scientific Diagram

Review of the Long-Term Implantable Senseonics Continuous Glucose Monitoring System and Other Continuous Glucose Monitoring Systems | Semantic Scholar
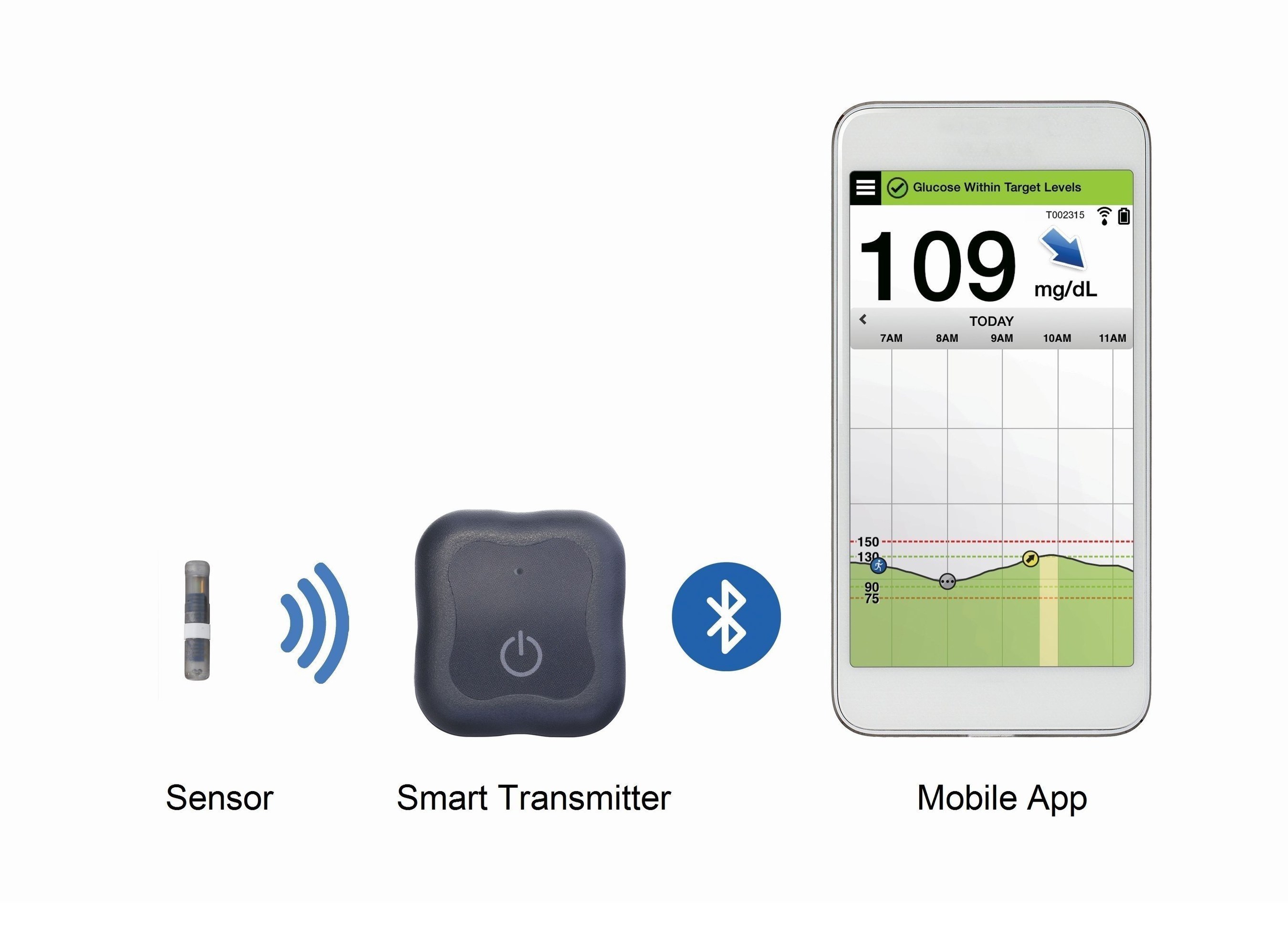
Roche Diabetes Care and Senseonics Announce Distribution Agreement for the Eversense® CGM System Introducing the First Implantable Long-term Glucose Sensor