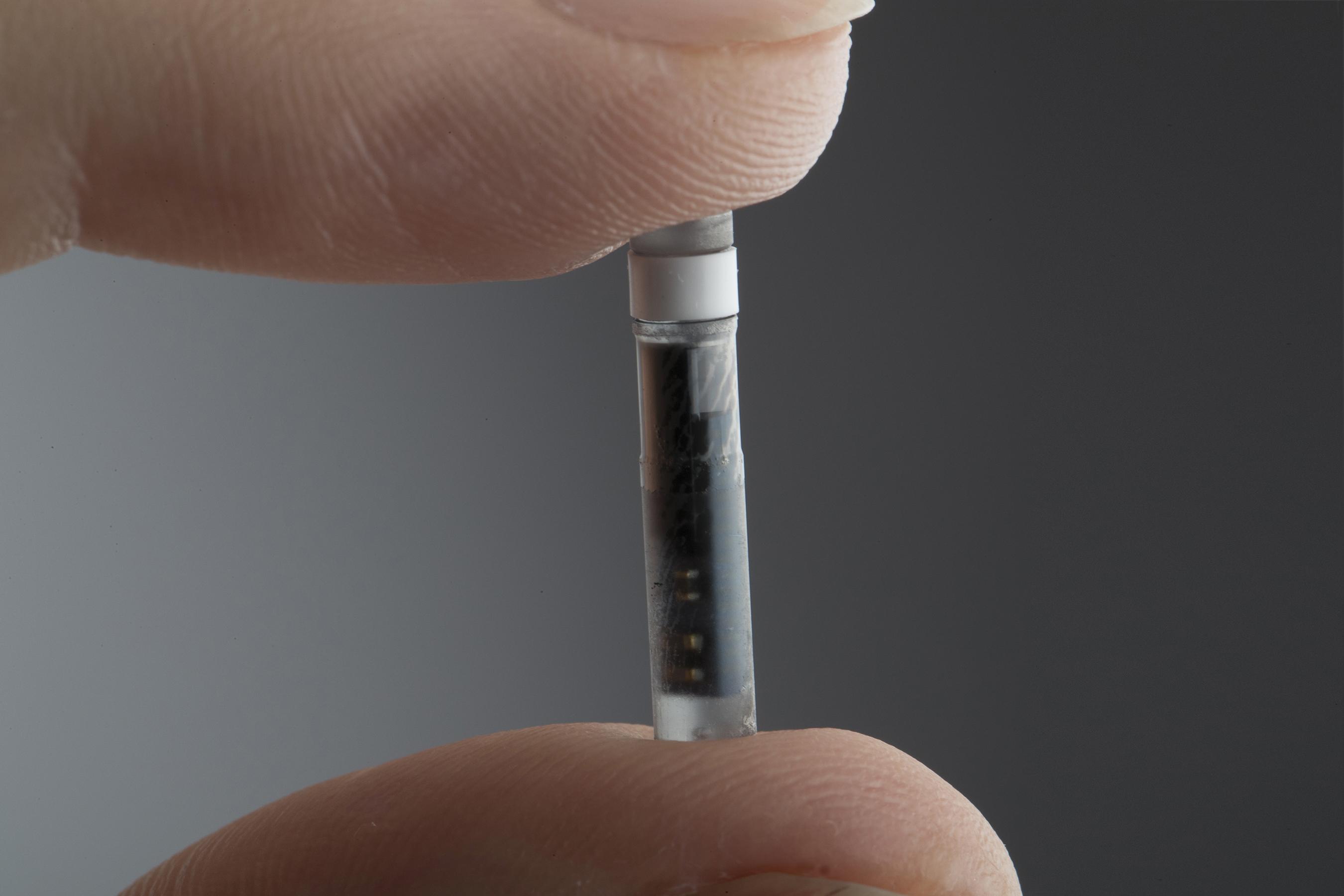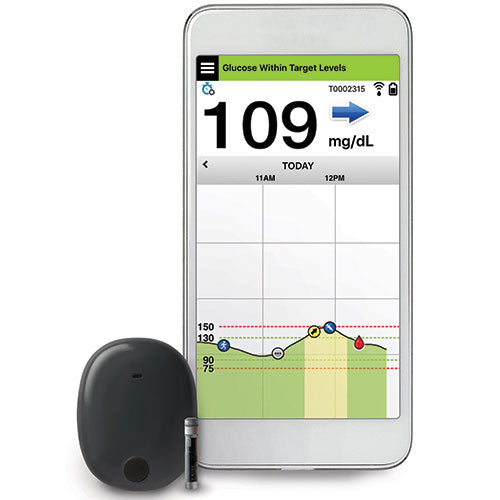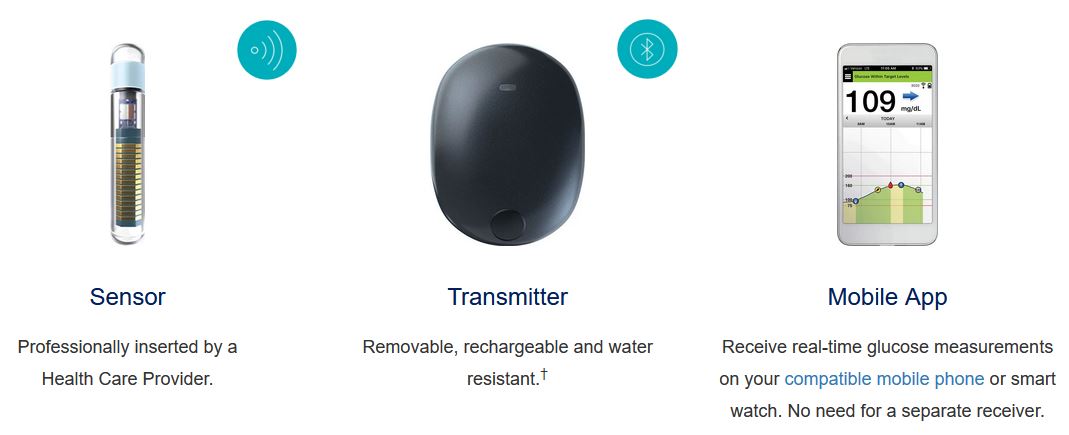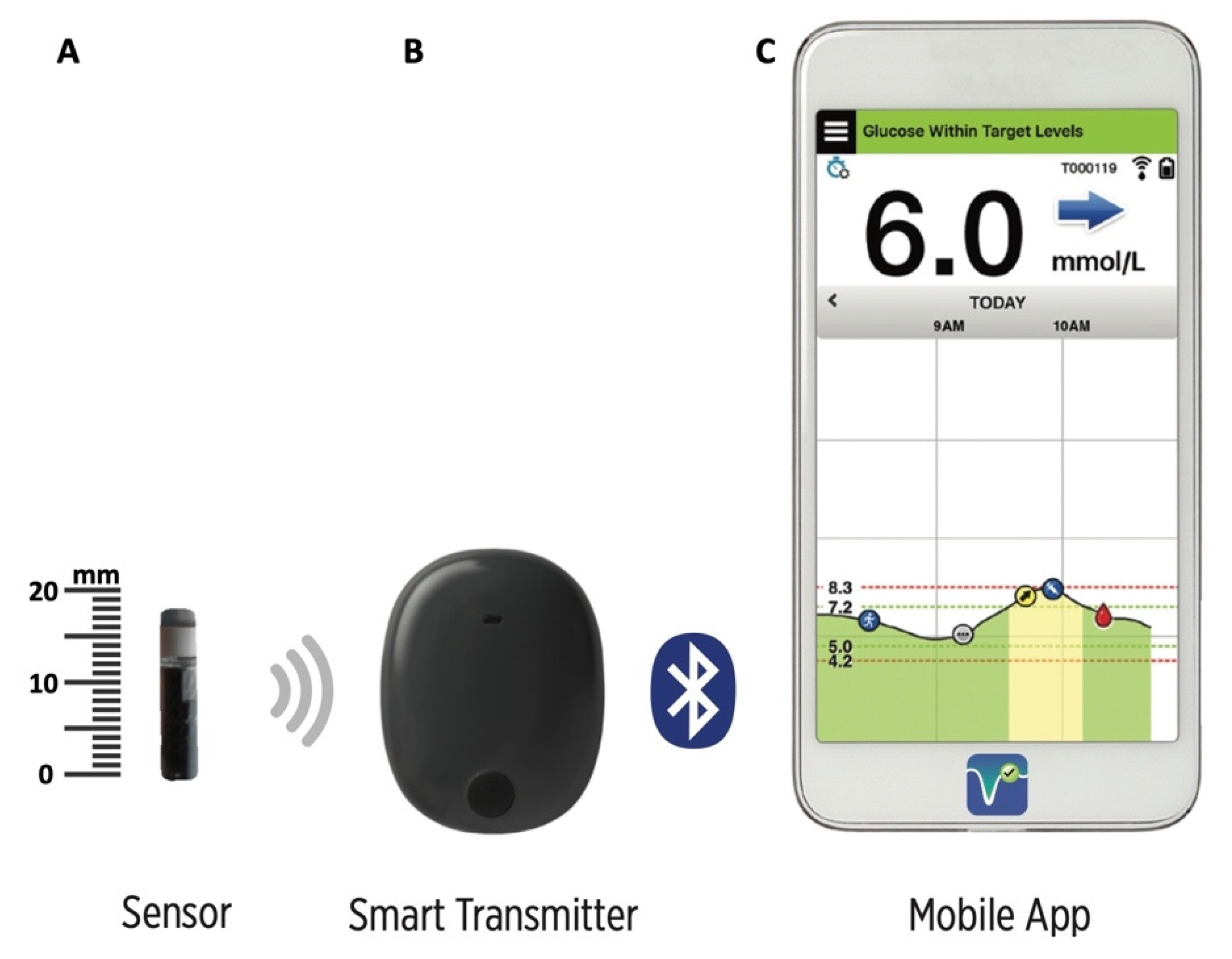
Animals | Free Full-Text | Clinical Use of a 180-Day Implantable Glucose Monitoring System in Dogs with Diabetes Mellitus: A Case Series

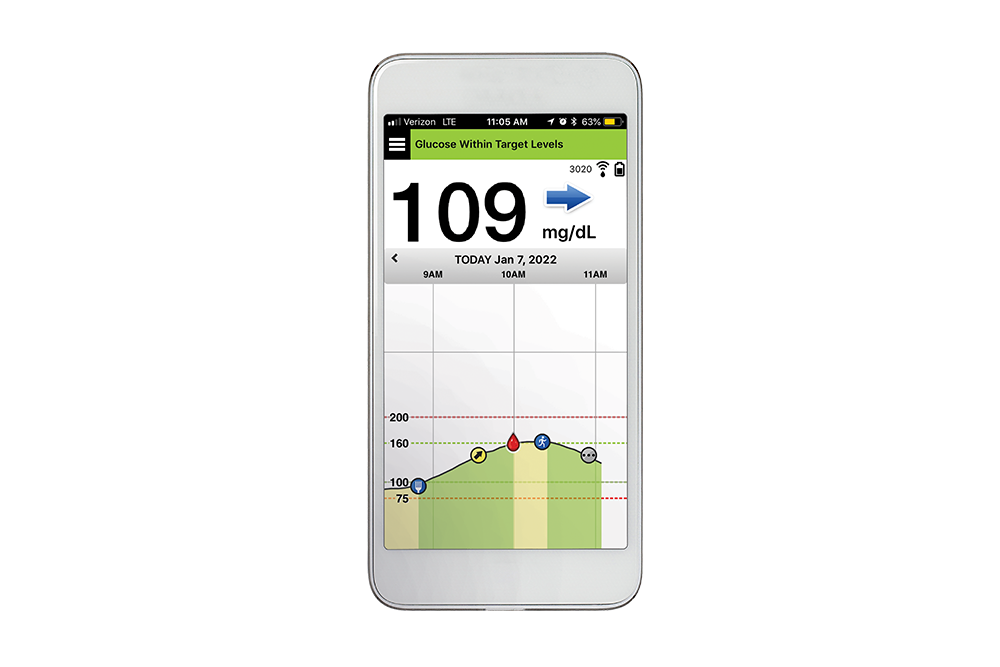
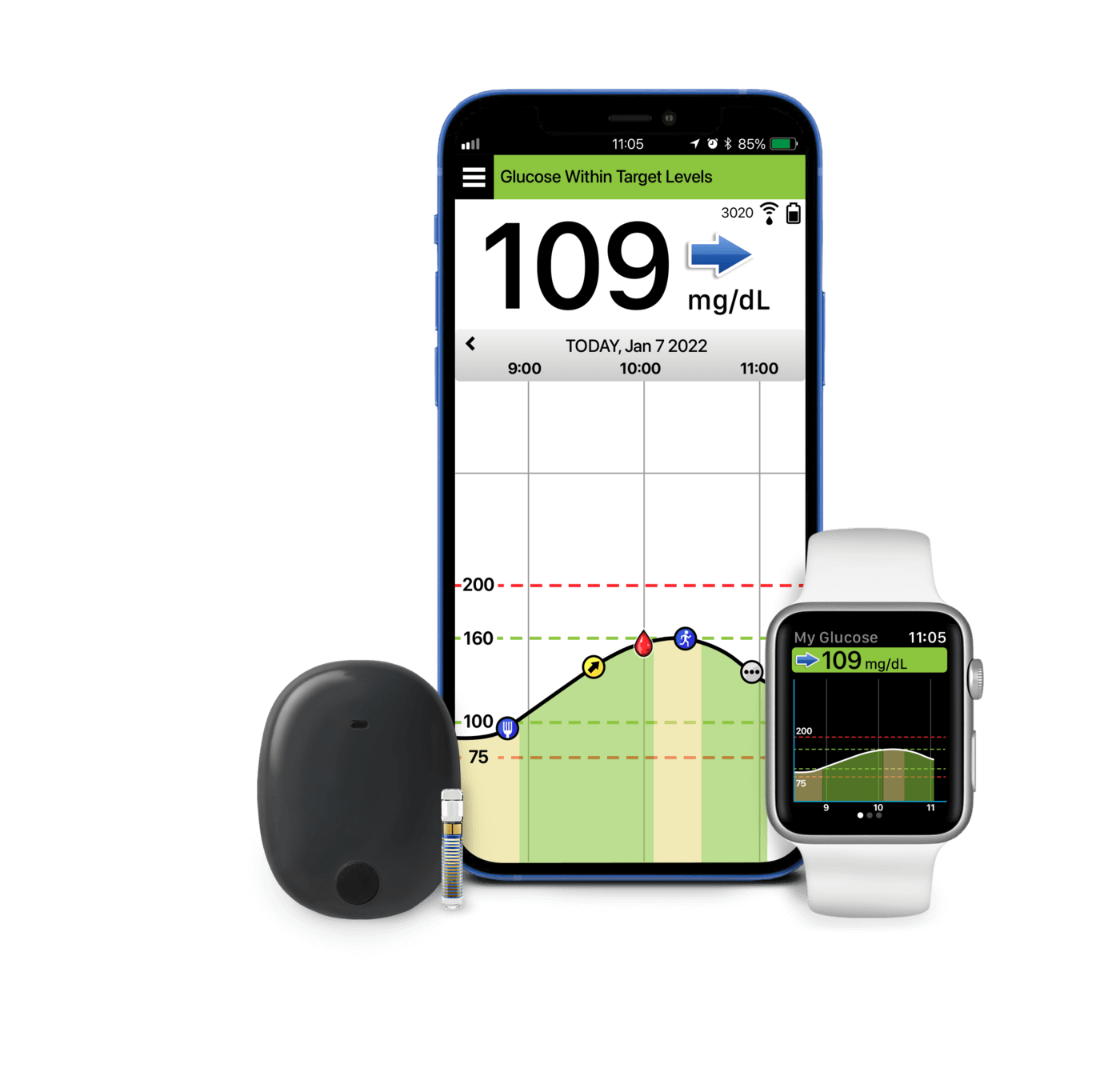
FDA approves Eversense E3 6-month continuous glucose monitor that requires fewer fingerstick blood glucose measurements - NotebookCheck.net News
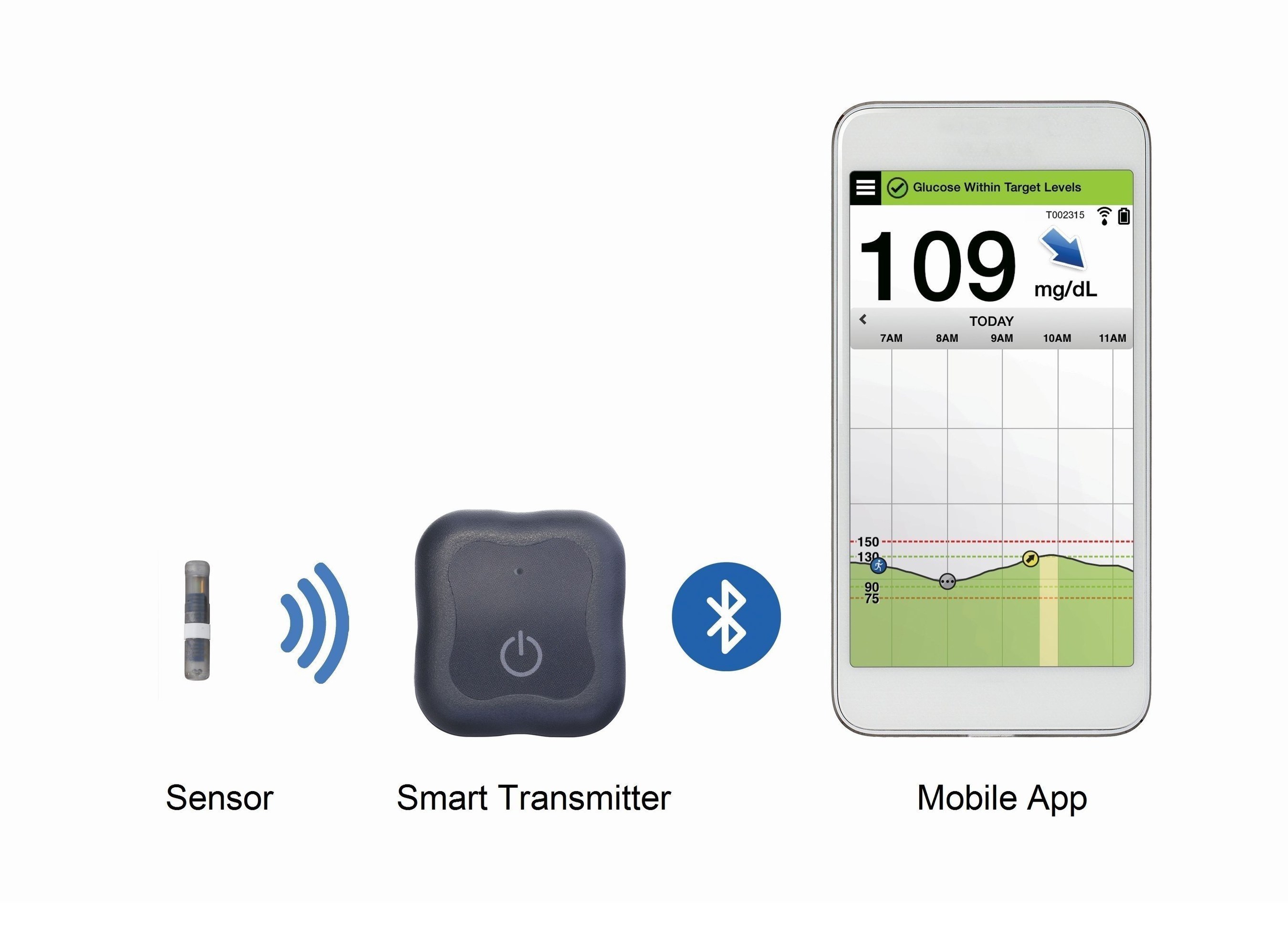
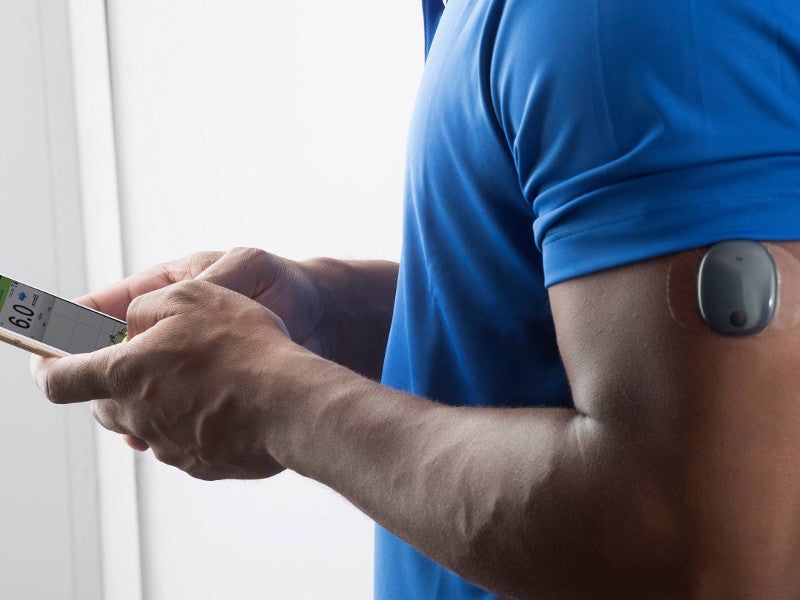
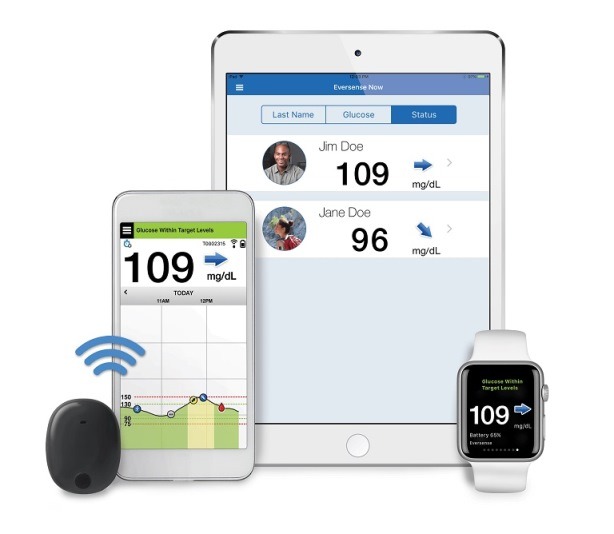
Roche Diabetes Care and Senseonics Announce Distribution Agreement for the Eversense® CGM System Introducing the First Implantable Long-term Glucose Sensor